 |
Oxygen, Carbon, Hydrogen
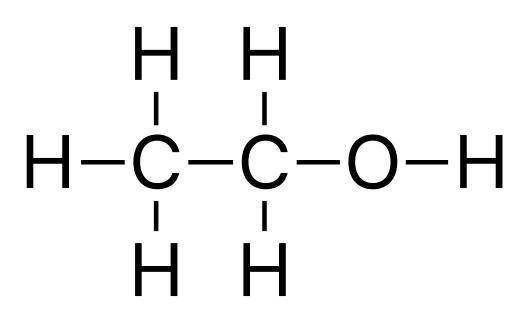
C2H6O, also known as Ethanol, is a polar molecule. You can see this from the 1st diagram, there is no clear line of symmetry. As you look at the 2nd diagram, you see the electronegativity of it’s atoms. The arrows point from the atoms with less electronegativity to the atoms with the higher electronegativity. As you can see, there are 3 Hydrogen atoms pointing to Carbon, 2 Hydrogen atoms pointing to the other Carbon, and a Carbon and Hydrogen atom pointing to the Oxygen atom. These arrows are simply depicting the electronegativity values within the molecule. Oxygen has an electronegativity value of 3.5 so Carbon, 2.5, and Hydrogen, 2.2, point to it. That is also why Hydrogen points to Carbon.
1.) The ad is very attractive and would make me want to buy your product, because ethanol is very useful.
ReplyDelete2.) Your model and diagram are both correct and well done, the arrows in the electronegativity are pointing the right way
3.)The appearance of your blog is appealing and easy to follow. The background draws attention to the text
4.) I agree with you that ethanol is polar, that is correct
5.) The intermolecular forces are correctly listed an well described also.
Divver...
ReplyDelete1.) The general appearance of your blog is easy on the eyes and the windmill in the background is ingeniously relevant and clever.
2.) The ad is attractive and entices the reader into wanting to buy the product. I, myself, now want to donate money into the research of ethanol as a possible fuel source.
3.) Your model and diagram seem to be correct and thoroughly done. The electronegativity arrows are pointing the right way and the atoms are clearly defined.
4.) Ethanol is definitely polar, so once again you were correct.
5.) The intermolecular forces are correctly defined and listed. The explanations are well done and scientifically accurate.
Good work!
Very nice diagram to explain the ethanol molecular structure.Its the basic molecule of alcohol which people drink.
ReplyDeletegeometry help